|
So here, we're showing theĮlectron being knocked loose and so the electron's moving in, let's just say, this direction, with some velocity, v, and if the electron has mass, m, we know that there's a kinetic energy. To hit this electron, and if the photon has enough energy, it can free the electron, right? So we can knock it loose, and so let me go ahead and show that. So this is massless, and the photon is going So I'm gonna draw in a particle of light which we call a photon, So this is kind of like a collision between two particles, if we think about lightĪs being a particle. Of those electrons loose, which causes a current The right kind of frequency, you can actually knock some If you shine a light on the metal, so the right kind of light with I'm gonna go ahead andĭraw one electron in here, and this electron is bound to the metal because it's attracted to the positive charges in the nucleus. So let's say you had a piece of metal, and we know the metal has electrons. And, an example of this, would be the Photoelectric effect,Īs described by Einstein. Sometimes light seems to act as a wave, and sometimes light seems And the electrons that are ejected would, indeed, come from the outermost shells but not because the photon would find it easier to remove them but because these are the electrons that are involved in the metallic bonding process and thus most available to the photon and any other such physical process. The work function (I would suggest) is not so much a function of the atom but more of the metal as a whole. 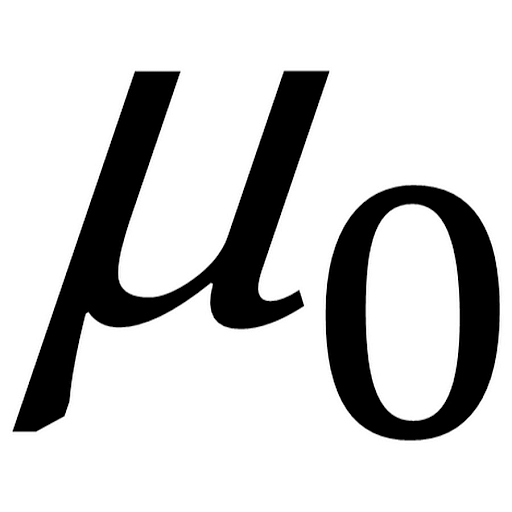
It will smash into this cloud or soup of electrons as they swirl around (or, more likely, wave around.) and give its energy to one of them. So, if this is still the current atomic model of a metal, I would say that your incident photon does not really get to see an atom as such. This is what carries the electric current and what makes metals such good conductors. 
the outermost electrons of each atom finds sufficient energy to leave its parent atom and joins other such electrons to 'move around' in a "sea of electrons". when I was younger, we used to talk about the structure of metals like this: "a metal consists of a regular array of positive ions. H or Planck's Constant is 6.26 * 10⁻³⁴ m² kg / secĮDIT: many thanks to Cipher for the correction! Λ(pronounced lambda) or the wavelength was given as 525 nanometers or 5.25 * 10⁻⁹ meters M or the mass (in this case an electron) is 9.11 * 10⁻³¹ kgĬ or the speed of light is 2.998 * 10⁸ meters / secondĮ₀ or Epsilon Naught was given as 3.43 * 10⁻¹⁹ Joules Now solve for velocity which is the v² in the above equation using the following numbers. This leaves us with ½*m*v² equals h*c/λ minus E₀ ½ * m * v² equals h* - E₀ because frequency(v) equals velocity of light(c) divided by the wavelength (λ) Note the substitution of c/λ where the frequency once was. ½ * m * v² equals minus E₀ because Epsilon photon equals h*v Note the substitution of h*v where Epsilon photon was. equals Epsilon photon minus E₀ because KE equals KE Kinetic Energy (KE) also equals Epsilon photon minus Epsilon naught (E₀) Kinetic Energy (KE) equals ½ * mass of electron (m) * the velocity(v)² The second issue is the format layout of how he got to his answer. 
On paper I always write my v's differently than my v's. Google "Greek alphabet" and view the wikipedia article to see what I mean. Both use the same character but represent different things. The first issue is v (the English alphabet character "ve") to represent velocity and v (the Greek alphabet character "nu") to represent frequency. Consider a sinusoidal electromagnetic wave with fields E ⃗ = E max ȷ ^ cos ( k x − ω t ) \vec B fields oscillate in phase.I also ran into the issue of wanting more explanation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
